|
In May of this year (2019), my lab group traveled to Green Island, Taiwan for a month long field season to culture planktic forams. This wasn't my first adventure to a field location to culture forams, but the first time I've traveled to such a remote location. Although Taiwan itself isn't exactly 'remote', Green Island is.. Travel required a car rental, 17 hour flight, a taxi ride, a stay in a hotel, a train ride, a taxi ride to a ferry, a 45 minute ferry ride, then another taxi to the scooter rental, then a scooter ride to the lab. In all it took about 48 hours until we arrived at the lab. Leaving for any emergency would take a similar amount of travel, which caused me an enormous amount of travel anxiety (future blog post on academic travel anxiety coming soon).. Every year that I've had a field season, I promise myself that I'm going to blog from the field. And every year I fail miserably at this effort (see my field blogging attempts in 2013, 2014, 2015). Days in the lab are sooooo veeeeery looooong. On Green Island, we'd get up at 5:30 am (sometimes 5 am to do yoga first, because #selfcare) and have the truck loaded to get to the dive boat by 6:30 am. We are out on the water by 7am at the latest and return most days 2-3 hours later (with the exception of our EPIC water sampling day). Then it's a frenzy of going through the dive material, plankton tow collections, AND we still need to observe and possibly feed every single foram we already have in culture. Each group has their own set of daily tasks for their forams and everyone pitches in to help things go smoothly. We took breaks for lunch, dinner, and often end up in the lab until late in the evening. Though many nights after dinner we'd have a beer or a T&T and then it'd be off to bed with sometimes little other work accomplished because we're all just SPENT. Rinse. Repeat. We DO have some breaks here and there, sometimes on Sundays (#SundayFunday), when we didn't do a dive/tow collection and then we'd have time to do a fun dive/snorkel, see some sights on the island (pics below!), and relax. But the time to blog is always hard to find. So this year, we tried something different. Instead of blogging HERE on my website, we did a group effort and blogged on the NatGeo Open Explorer forum: https://openexplorer.nationalgeographic.com/expedition/huntfortheforams Most of the posts were written by CEOAS graduate students Kelsey Lane, who did an AMAZING job! There were also a few posts by Theresa Fritz-Endres, a post by Oscar Branson who blogged about diving for forams and a few posts by me. This was by far a way more successful blog than we've ever had in terms of the number of blog posts. The downside about the NatGeo blog format: You can only post 4 photographs per post and at the END of your post unless you store them somewhere else online (GitHub, Flickr, etc). Because of this, a few of our posts still need photographs (coming soon I hope!). But all in all, NatGeo's Open Explorer was a GREAT way to blog for a field season and I highly recommend it if you're looking for a great avenue for blocking about your field research adventures. Published 10am on 1 Sept., 2019 Update 11am on 1 Sept., 2019: I just found out NatGeo is no longer supporting their OpenExplorer blog. I admit I'm a little sad to hear that this amazing platform will not longer be supported. A new platform will take its place at National Geographic Society for NGS-supported expeditions. So, onward to a new finding a new blogging location for our next field campaign.
3 Comments
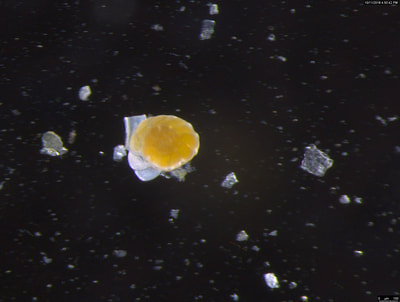
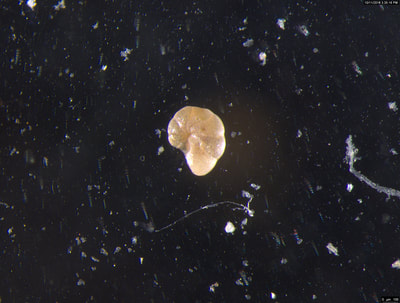
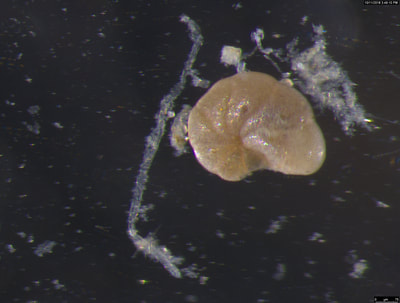
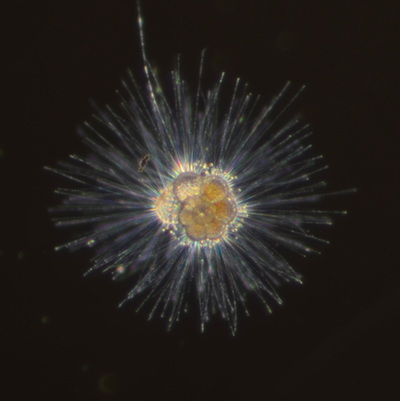
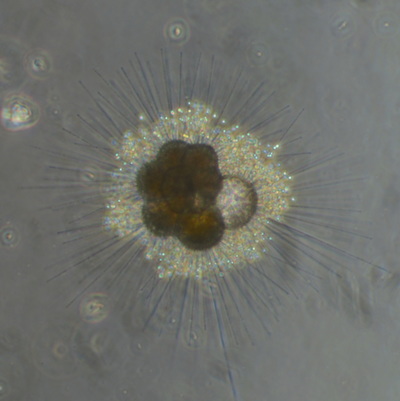
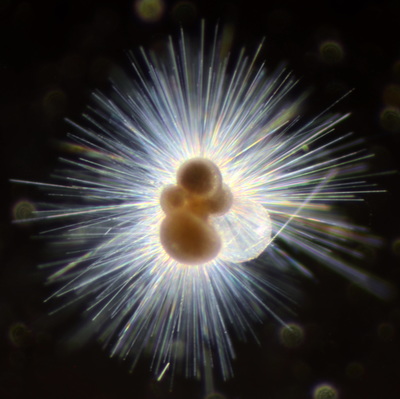
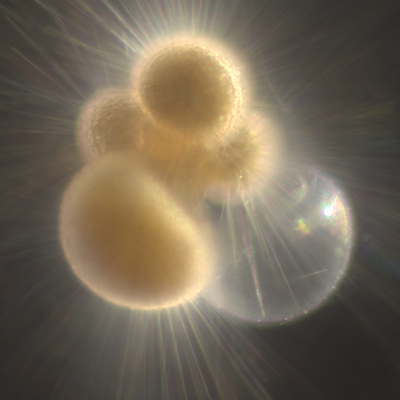
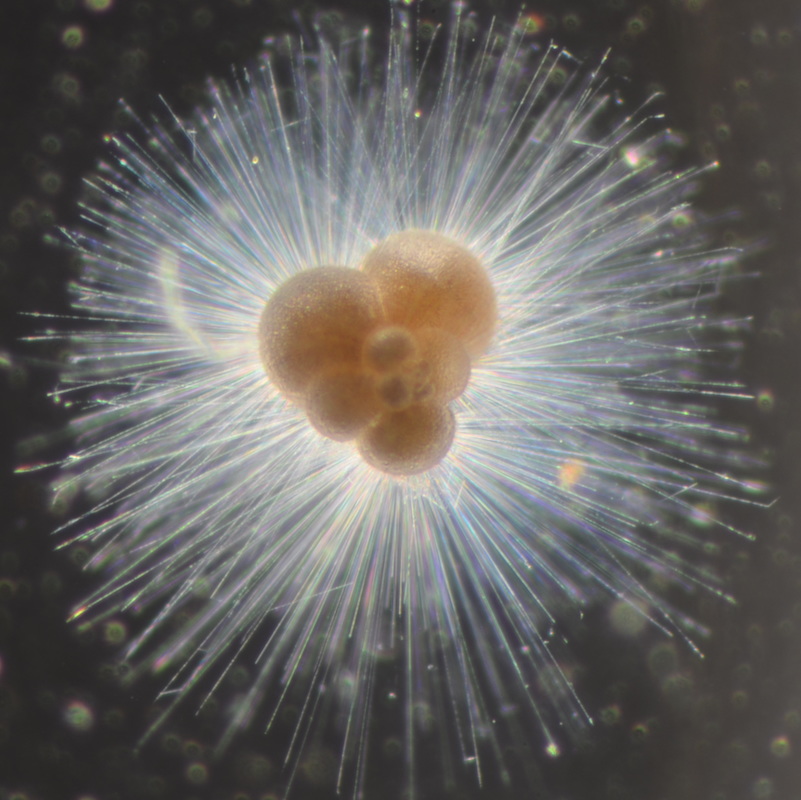
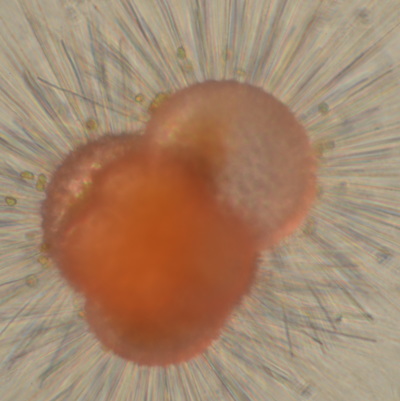
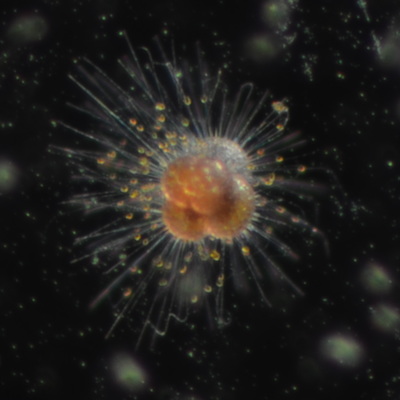
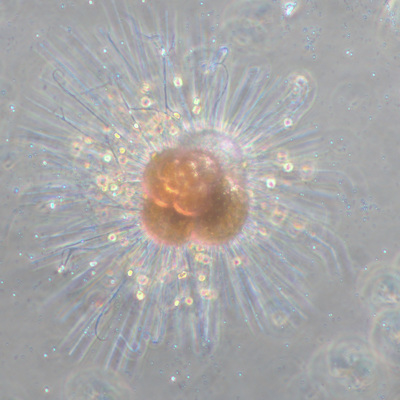
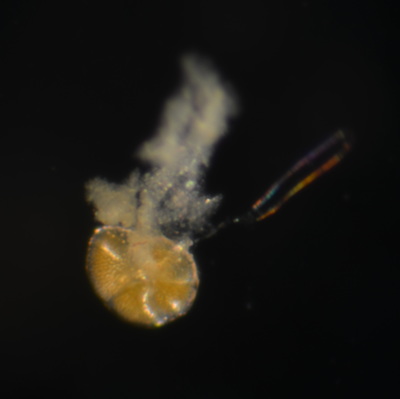
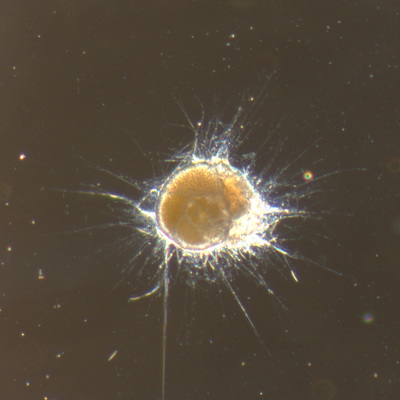
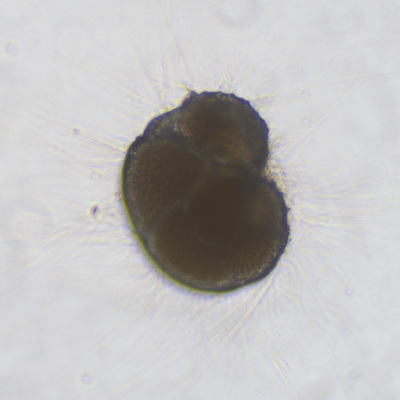
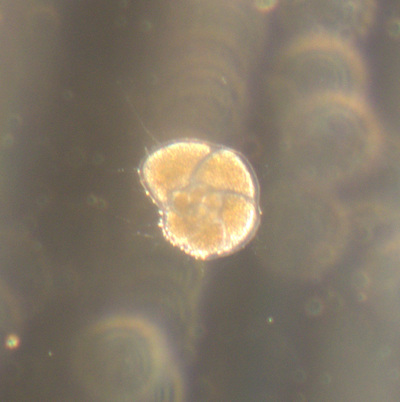
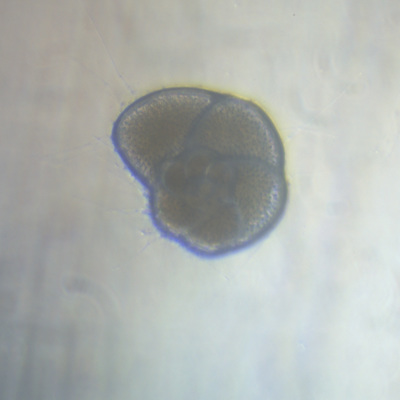
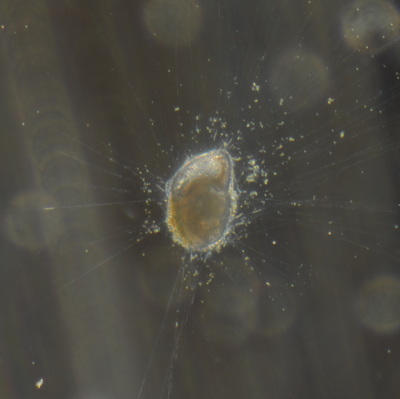
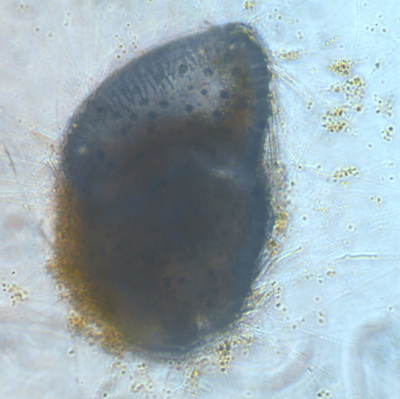
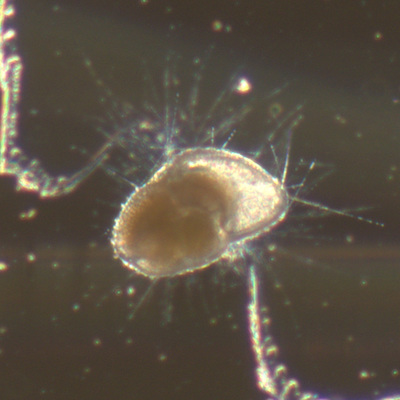
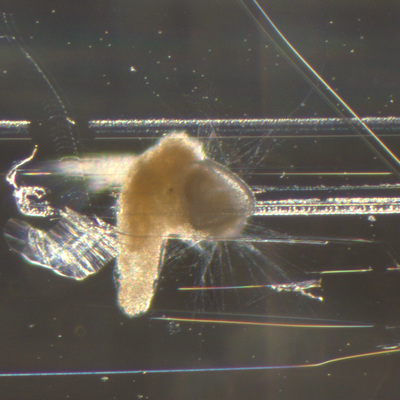
I've been at Oregon State University for just over 2 years. My lab has been finished for just over a year, I have 2 graduate students now, and have hosted 6 undergraduate researchers in my lab. In the last few years, I've been out to sea a few times to collect planktic forams to begin to test the feasibility of culturing planktics here. It definitely can be done, though it is more challenging compared to culturing at a place like, for instance, Catalina Island, where the seas are calm most mornings and the foram species available are culture friendly (there is a reason we refer to O. universa as the lab rat). I've also been tinkering with the idea of culturing benthic forams local to this region. But first, I had to find some. And so last week we headed to the coast in the early morning hours to dig around in the muddy tidal flats for benthic forams. My colleague, George Waldbusser, took us to a few places he's familiar with. We started near the Hatfield Marine Science Center (photo on left below) and then headed to Sally's Bend (photo on right) a bit south of Newport, Oregon. It was a gorgeous morning for field sampling: No rain, no wind, and clear skies! We sampled our first site just as the sun started to rise. You'll notice the rookie professor on the left in the photos above. Yep, that's me. In not so great field gear. (ok, so that isn't anywhere close to field gear). I recently lent my waterproof boots and bibs to a student and forgot they were in a bin in the lab instead of a bin in my garage. So I woke up at 5am, basically ready to go (car packed the night before, snacks, first aid kit, coffee, tea in a thermos...) but without any field gear. I thought, well, it's going to be a calm warm morning, I'll just pack extra cloths in case I need them... So there I am, in the mud flats, with my properly dressed students, and I'm in jeans and a decades old pair of boots (UGH! such a rookie). This was definitely a teaching moment: Do as I say not as I do! Do not attempt. (I will never do this again...). At least I packed the extra boots and clothes to change into. Which were pretty useful after we sank in the mud at Sally's Bend and fell over trying to get out. Post sampling, we ate at an amazing breakfast place in Newport (La Maison), and then headed back to the lab to start picking for forams. The foram search was slow, but we did find live calcareous benthics (the agglutinated species are more common). So despite the cold toes and lesson learned (Don't be like Jenn. Have extra foul weather gear and boots!), we did find live forams (and some dead one's)! It was a successful hunt! (and damn, those benthic forams sure are pretty...) We just finished a 'short' (5 week) field season for culturing planktic foraminifera at the Wrigley Marine Science Center on Catalina Island. This is our 4th field season in 5 years and the last field season on our current culture grant. We LOVE coming here. We can capture live forams less than 10 minutes from the lab in deep water and have them in culture within hours of collection. This summer, mother nature did not cooperate with our research plans. Instead of getting the usual species we have seen here over the last few years, mother nature threw us a curve ball and El Nino is to blame....The water mass off the coast of Catalina averaged 22.5°C over the course of our field season (nearly 24°C on some days) and was very oligotrophic. Probably sub-tropical in origin... Not the water mass we need if we want the nutrient loving N. dutertrei (or at least we think it is nutrient loving). We needed to collect O. universa and N. dutertrei. G. bulloides would have been ideal too for our collaborators. We did manage to culture enough O. universa and N. dutertrei to complete most of our experiments, but we really needed numbers to be higher. G. bulloides was nearly absent. So what did we get? Forams that are not common here: G. humulis (I didn't even know of this foraminifer until this summer - I've never seen it in fossil samples I've picked through), G. calida, T. rubescens, G. siphonifera, a few Globorotalids and a handful of others... It wasn't ideal, but we made the best of it by getting some specimens we caught in the nets to recover or taking dive caught specimens and growing them in seawater labeled with an isotope so we could track calcification. And of course, we took pictures and documented what we learned. Here are a few of my favorites... Globigerinita humilis This is one tiny foram. This one measured <150 microns on the longest dimension. It had very light brown cytoplasm (darker on the inverted scope). The first day it was caught it did not have symbionts out and the spines were nearly absent and the last two chambers were very thinly calcified and were not full of cytoplasm. These pictures were taken after day 5 in the lab. I did feed it a frozen (thawed) Artemia nauplii on day 2 or 3. Globigerinella calida This was a dive caught specimen. On day 2 or 3 in culture, it formed a new chamber, so we transferred it into seawater that contained an isotope label so that we can identify the calcite that grew in culture. It took us a LONG time to identify what species it was, because it was SO very weird looking in culture. The first two pictures were taken on day 2 or 3 and the last picture was taken on day 5 in culture. After the new chamber filled in, the specimen died or went through gametogenesis. Globigerina rubescens I really don't know if the name above is correct: is it a Globoturborotalita rubescens or Globigerina rubescens. Either way, I'm certain we have never seen this in the plankton tows here on Catalina Island. They are SO distinctly different from all of the other forams because they are quite small and their shells are VERY pink in color. Almost like the pink G. ruber. They were very very abundant in our tows - probably the most abundant of all of the forams. The divers never caught one of these, probably because they are so small. Or they don't live at the surface where the divers spend heir time (<5m depth). We tried to get these to recover and grow in seawater with an isotope label, but very few recovered. Only two recovered well enough to be photographed. Globorotalia menardii G. menardii has been in culture before by other researchers, but we don't often capture them near Catalina Island. This one has been in culture since it was caught on August 13 and it is still kicking. On occasion, it looked very deformed as it grew and added chambers, but then would look like it calcified without any deformation, so I'm not really sure how to interpret what we've observed on a day to day basis. Globorotalia crassaformis (?) This deep dweller didn't live very long and is archived so we can take pictures on the SEM. We think it is a G. crassiformis. G. crassaformis or truncatulinoides Initially identified as a G. truncatulinoides... The jury is still out on this one. Our field season overlapped with a workshop on the biology of planktic foraminifera and several researchers observed this specimen. Two thought it was a G. crassiformis and two thought it was a G. truncatulinoides. I think post-culture SEMs will be needed. It was captured on 8/26 and is still alive. It was transferred in and out of labeled seawater on a day/night schedule to label calcification on a 24 hour time period for 9 days. Then on 9/5 it was transferred into a different label where it will stay until it undergoes gametogenesis or dies. The first two pictures were taken the day after this specimen was collected. The next two pictures were taken 7 days after collection (one after it was fed an artemia). Notice the small brown specks around the foram in the first two pictures? I have no idea what those are but they look REALLY similar to the symbionts found around N. dutertrei. According to Gastrich, 1987, G. truncatulinoides doesn't contain symbionts. I'm not sure anyone has investigated the presence/absence of symbionts in G. crassiformis. In any case, we aren't sure what these specks are. We do know that N. dutertrei expels its symbionts a few days before gametogenesis, so perhaps some of the other deep dwellers have symbionts and the ones investigated thus far were pregametogenic adults without symbionts? Just a thought.
|
Proudly powered by Weebly













 RSS Feed
RSS Feed